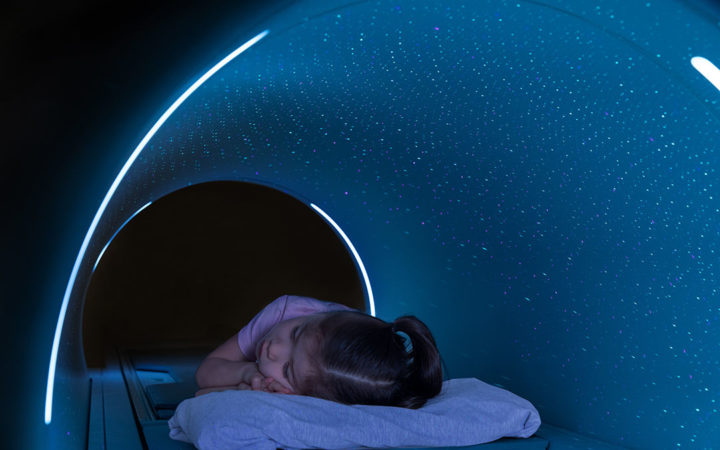
In May, United Imaging received FDA clearance for its uMR OMEGA MR scanner, a system equipped with the world's first ultra-wide 75-cm bore.
"There's a lot about this product that fits patients in the United States," said Jeffrey Bundy, Ph.D., chief executive officer of United Imaging Healthcare Solutions. "Clearly one of the things patients talk about is the size of the scanner."
The 3T scanner has a 680-pound capacity and includes a larger area of homogeneous signal and magnetic field to image larger patients and allow them to be more comfortable.
"If you're just going to quote specifications in one area or the other, maybe it doesn't rise to being a solution," Bundy said. "That solution requires the image quality to be better, which is accomplished primarily with the significant improvement in magnet homogeneity."
The signal-to-noise ratio in a 3T scanner is better than it would be in an open MR, which has a lower magnet field strength."
With greater comfort, there may be less of a need to sedate pediatric patients, and less of a need to reschedule patients who aren't able to undergo an exam due to discomfort.
In the MR technology space, last year, the company released a compressed sensing package called United Compressed Sensing, or uCS for all 1.5T and 3T scanners.
Earlier this year, the company received FDA clearance for its EasyScan Cardiac, a fully-automated cardiac localizer tool, for the OMEGA and the uMR 570.
"We believe uMR OMEGA will help our customers scan more patients and get much more flexibility out of their capital investment, which is even more important now than it was just a few months ago," Bundy said.