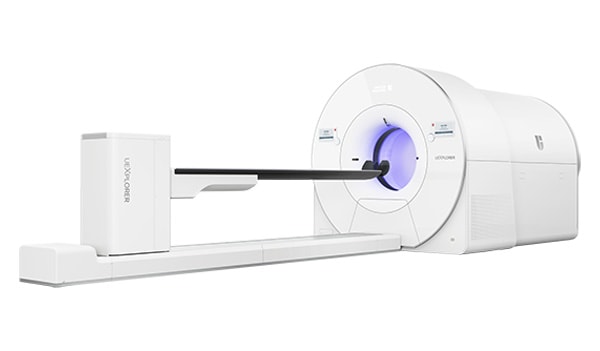
Telix and BAMF Health use Illuccix® (kit for the preparation of Gallium Ga 68 gozetotide injection) for the first time with uEXPLORER® total-body PET scanner for the detection of prostate cancer
GRAND RAPIDS, Michigan and INDIANAPOLIS, Indiana – July 27, 2022 – In a combination never before used to scan men diagnosed with prostate cancer, the PSMA-PET1 imaging agent Illuccix® from Telix Pharmaceuticals Limited (Telix) was used in patients this week with uEXPLORER®, a first-of-its kind total-body PET scanner from United Imaging. Nine men with prostate cancer underwent the procedure at BAMF Health (Bold Advance Medical Future) on the "Medical Mile" in Grand Rapids as part of an investigator-led study.
The uEXPLORER is the world's first and only medical imaging 3D scanner capable of capturing the total human body in a single bed position and allows physicians to conduct a total-body PET scan in less than 10 minutes – compared to approximately 40 minutes in a conventional PET/CT. The scan may potentially open significant testing capacity for physicians, with some medical facilities able to image up to 40 patients per day2 with uEXPLORER.
BAMF Health is a leading "theranostics" (therapeutic and diagnostic) cancer treatment center that delivers precision medicine through artificial intelligence-enabled molecular imaging and theranostics. Telix launched Illuccix for prostate cancer imaging in April in the U.S. and has a pipeline of imaging and therapeutic agents in development to potentially detect and treat a range of cancers and rare diseases through targeted radiation.
Illuccix is a radioactive diagnostic agent indicated for imaging of Prostate-Specific Membrane Antigen (PSMA) via positron emission tomography (PET) indicated for positron emission tomography (PET) of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer:
- with suspected metastasis who are candidates for initial definitive therapy
- with suspected recurrence based on elevated serum prostate-specific antigen (PSA) level.
PSMA-PET is a diagnostic tool clinically demonstrated to detect advanced diseases that may not appear in conventional imaging3 and is now included in key clinical practice guidelines for prostate cancer, such as the NCCN Clinical Practice Guidelines in Oncology.4
The molecular imaging and molecular therapy clinics at the BAMF Health Theranostics Center include an on-site dual cyclotron-equipped radiopharmacy. Dr. Matt DeLong, vice president, radiopharmacy at BAMF Health, said, "We are beyond excited to have been able to prepare and dispense the Illuccix doses on demand. Illuccix allows us to leverage the capability of the cyclotrons and generators within the radiopharmacy."
The uEXPLORER, which is manufactured by global medical imaging equipment provider United Imaging, includes increased geometric coverage of the body and improved timing resolution, thereby increasing sensitivity and reducing the time of a scan from 40 minutes to less than 10 minutes.5
Dr. Christian Behrenbruch, Group CEO and Managing Director of Telix Pharmaceuticals said, "This is an important opportunity for patients and medical teams alike. PSMA-PET imaging has been one of the most important developments in prostate cancer detection and nuclear medicine in recent years – and is already being rapidly adopted by medical teams managing men with prostate cancer. We're excited to explore this approach to PSMA-PET imaging with BAMF Health and to demonstrate our commitment to ongoing innovation in this field."
Dr. Harshad Kulkarni, BAMF Health Chief Medical Advisor, said, "At BAMF Health we are focused on bringing new theranostic technologies and treatment options to patients in the United States. This partnership brings together imaging hardware technology and radiopharmaceutical developments. The uEXPLORER has the potential to offer increased sensitivity and faster scan time. These efficiencies may help get patients vital information about their cancer more quickly and allow us to see more patients in need. The uEXPLORER could be a difference-maker for medical teams who need to be able to treat more patients and get results quickly, and for patients who want the comfort of less time in a PET/CT."
Prostate cancer is the second-most common cancer among men in the U.S. According to the American Cancer Society, more than 268,000 men in the U.S. will be affected this year by prostate cancer, and nearly 35,000 will die from the disease. Reliable and flexible diagnostic tools are essential for treatment teams in narrowing the gap between accurate staging and appropriate individualized treatment by health care professionals.
About Illuccix
Telix's lead product, Illuccix® (kit for preparation of gallium-68 (68Ga) gozetotide (also known as 68Ga PSMA-11) injection) for prostate cancer imaging, has been approved by the U.S. Food and Drug Administration (FDA),6 and by the Australian Therapeutic Goods Administration (TGA).7 Telix is also progressing marketing authorization applications for this investigational candidate in Europe8 and Canada.9
Illuccix®, after radiolabeling with Ga 68, is a radioactive diagnostic agent indicated for positron emission tomography (PET) of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer:
- with suspected metastasis who are candidates for initial definitive therapy
- with suspected recurrence based on elevated serum prostate-specific antigen (PSA) level
About Telix Pharmaceuticals Limited
Telix is a biopharmaceutical company focused on the development and commercialisation of diagnostic and therapeutic products using Molecularly Targeted Radiation (MTR). Telix is headquartered in Melbourne, Australia with international operations in Belgium, Japan, Switzerland, and the United States. Telix is developing a portfolio of clinical-stage products that address significant unmet medical need in oncology and rare diseases. Telix is listed on the Australian Securities Exchange (ASX: TLX). For more information visit www.telixpharma.com and follow Telix on Twitter (@TelixPharma) and LinkedIn.
About BAMF Health
BAMF Health is achieving Intelligence-Based Precision Medicine through Artificial Intelligence enabled Molecular Imaging and Molecular Targeted Radiation Therapy. The company's flagship location in Grand Rapids, Michigan will include the world's most advanced cyclotron-equipped Radiopharmacy, imaging clinic, and Theranostic clinic, and will open inside of the Doug Meijer Medical Innovation Building in Summer 2022. To learn more, visit bamfhealth.com or follow us on LinkedIn.
About United Imaging
At United Imaging, we develop and produce advanced medical products, digital healthcare solutions, and intelligent solutions that cover the entire process of imaging diagnosis and treatment. Founded in 2011, our company has subsidiaries and R&D centers across the world. Our North American headquarters in Houston includes our corporate offices, factory, product showroom, service training center, and service parts distribution center. With a cutting-edge digital portfolio and a mission of Equal Healthcare for All™, and with innovative purchasing programs like All-in Configurations™ and Software Upgrades for Life™, we help drive industry progress and bold change. To learn more, visit united-imaging.com or follow us on LinkedIn and Twitter @UnitedImagingHC.
Important Safety Information Warnings and Precautions
Risk for Misdiagnosis
Image interpretation errors can occur with gallium Ga 68 gozetotide PET. A negative image does not rule out the presence of prostate cancer and a positive image does not confirm the presence of prostate cancer. The performance of gallium Ga 68 gozetotide for imaging of biochemically recurrent prostate cancer seems to be affected by serum PSA levels and by site of disease. The performance of gallium Ga 68 gozetotide for imaging of metastatic pelvic lymph nodes prior to initial definitive therapy seems to be affected by Gleason score. Gallium Ga 68 gozetotide uptake is not specific for prostate cancer and may occur with other types of cancer as well as non-malignant processes such as Paget's disease, fibrous dysplasia, and osteophytosis. Clinical correlation, which may include histopathological evaluation of the suspected prostate cancer site, is recommended.
Radiation Risks
Gallium Ga 68 gozetotide contributes to a patient's overall long-term cumulative radiation exposure. Long-term cumulative radiation exposure is associated with an increased risk for cancer. Ensure safe handling to minimize radiation exposure to the patient and health care workers. Advise patients to hydrate before and after administration and to void frequently after administration.
Adverse Reactions
The safety of gallium Ga 68 gozetotide was evaluated in 960 patients, each receiving one dose of gallium Ga 68 gozetotide. The average injected activity was 188.7 ± 40.7 MBq (5.1 ± 1.1 mCi). No serious adverse reactions were attributed to gallium Ga 68 gozetotide. The most commonly reported adverse reactions were nausea, diarrhea, and dizziness, occurring at a rate of < 1%.
Drug Interactions
Androgen deprivation therapy and other therapies targeting the androgen pathway
Androgen deprivation therapy (ADT) and other therapies targeting the androgen pathway, such as androgen receptor antagonists, can result in changes in uptake of gallium Ga 68 gozetotide in prostate cancer. The effect of these therapies on performance of gallium Ga 68 gozetotide PET has not been established.
YOU ARE ENCOURAGED TO REPORT SUSPECTED ADVERSE REACTIONS OF PRESCRIPTION DRUGS TO THE FDA.
VISIT MEDWATCH AT WWW.FDA.GOV/MEDWATCH OR CALL 1-800-FDA-1088. You may also report adverse reactions to Telix by calling 1-844-455-8638 or emailing: pharmacovigilance@telixpharma.com.
PLEASE NOTE THIS INFORMATION IS NOT COMPREHENSIVE. PLEASE SEE FULL PRESCRIBING INFORMATION AT ILLUCCIX.COM
Telix Investor Relations
Ms. Kyahn Williamson
Telix Pharmaceuticals Limited
SVP Corporate Communications and Investor Relations
Email: kyahn.williamson@telixpharma.com
Telix Media Relations
Mr. Greg Kueterman
Email: gkueterman@gmail.com
1 Prostate specific membrane antigen / positron emission tomography
2 Cherry et al, The Journal of Nuclear Medicine, 2018, Vol 59, No 1
3 Kase, A, et al The Continuum of Metastatic Prostate Cancer: Interpreting
PSMA PET Findings in Recurrent Prostate Cancer Cancers 2022, 14, 1361.
4 NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Prostate Cancer V.3.2022. Available at:
https://www.nccn.org/guidelines/category_1.
5 Cherry et al, The Journal of Nuclear Medicine, 2018, Vol 59, No 1
6 ASX disclosure 20 December 2021.
7 ASX disclosure 2 November 2021.
8 ASX disclosure 10 December 2021.
9 ASX disclosure 16 December 2020.
This announcement has been authorised for release the disclosure committee of Telix Pharmaceuticals Limited.
Legal Notices
This announcement may include forward-looking statements that relate to anticipated future events, financial performance, plans, strategies or business developments. Forward-looking statements can generally be identified by the use of words such as "may", "expect", "intend", "plan", "estimate", "anticipate", "outlook", "forecast" and "guidance", or other similar words. Forward-looking statements involve known and unknown risks, uncertainties and other factors that may cause our actual results, levels of activity, performance or achievements to differ materially from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. Forward-looking statements are based on the Company's good-faith assumptions as to the financial, market, regulatory and other risks and considerations that exist and affect the Company's business and operations in the future and there can be no assurance that any of the assumptions will prove to be correct. In the context of Telix's business, forward-looking statements may include, but are not limited to, statements about: the initiation, timing, progress and results of Telix's preclinical and clinical studies, and Telix's research and development programs; Telix's ability to advance product candidates into, enrol and successfully complete, clinical studies, including multi-national clinical trials; the timing or likelihood of regulatory filings and approvals, manufacturing activities and product marketing activities; the commercialisation of Telix's product candidates, if or when they have been approved; estimates of Telix's expenses, future revenues and capital requirements; Telix's financial performance; developments relating to Telix's competitors and industry; and the pricing and reimbursement of Telix's product candidates, if and after they have been approved. Telix's actual results, performance or achievements may be materially different from those which may be expressed or implied by such statements, and the differences may be adverse. Accordingly, you should not place undue reliance on these forward-looking statements. You should read this announcement together with our risk factors, as disclosed in our most recently filed reports with the ASX or on our website.
To the maximum extent permitted by law, Telix disclaims any obligation or undertaking to publicly update or revise any forward-looking statements contained in this announcement, whether as a result of new information, future developments or a change in expectations or assumptions.
The Telix Pharmaceuticals and Illuccix name and logo are trademarks of Telix Pharmaceuticals Limited and its affiliates
(all rights reserved).